Chemdoodle delocalized benzene12/29/2023  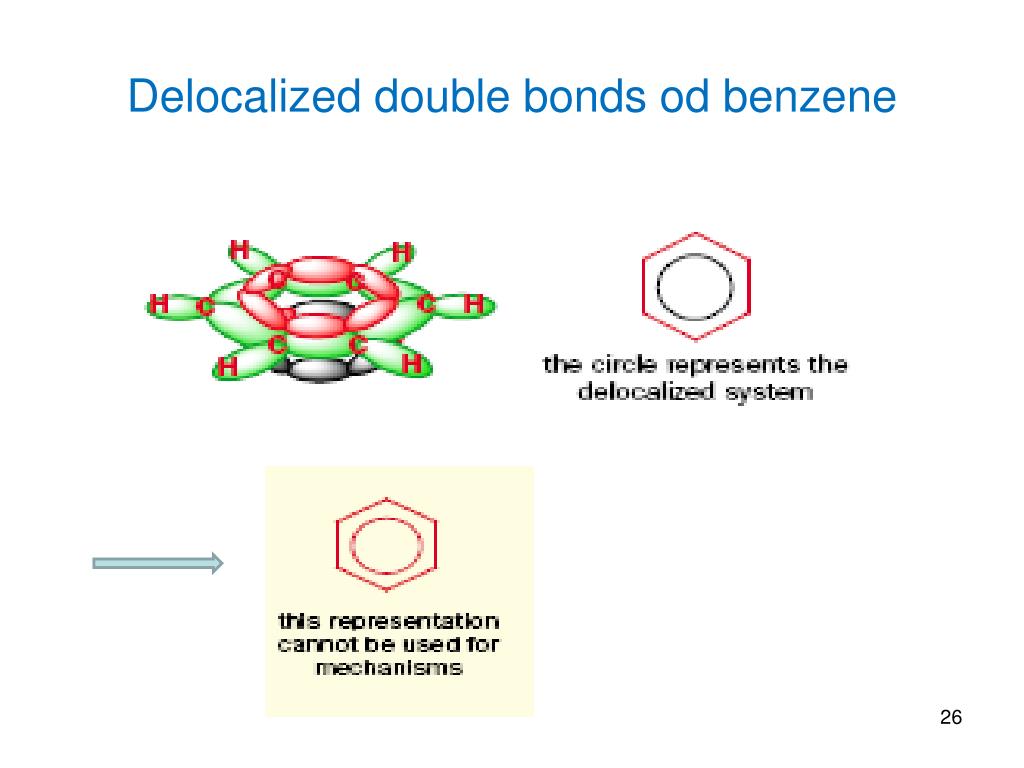 
The six-membered ring in benzene is a perfect hexagon (all carbon-carbon bonds have an identical length of 1.40 Å). Here, two structurally and energetically equivalent electronic structures for a stable compound are written, but no single structure provides an accurate or even an adequate representation of the true molecule. Eventually, the presently accepted structure of a regular-hexagonal, planar ring of carbons was adopted, and the exceptional thermodynamic and chemical stability of this system was attributed to resonance stabilization of a conjugated cyclic triene. The conceptual contradiction presented by a high degree of unsaturation (low H:C ratio) and high chemical stability for benzene and related compounds remained an unsolved puzzle for many years. This further confirms the previous indication that the six-carbon benzene core is unusually stable to chemical modification. If benzene is forced to react by increasing the temperature and/or by addition of a catalyst, It undergoes substitution reactions rather than the addition reactions that are typical of alkenes. As experimental evidence for a wide assortment of compounds was acquired, those incorporating this exceptionally stable six-carbon core came to be called "aromatic". For comparison, reactions of cyclohexene, a typical alkene, with these reagents are also shown (green box). Benzoic acid was eventually converted to the stable hydrocarbon benzene, C 6H 6, which also proved unreactive to common double bond transformations, as shown below. The H:C ratio in benzoic acid is <1, again suggesting the presence of several double bonds. On treatment with hot permanganate solution, cinnamaldehyde gave a stable, crystalline C 7H 6O 2 compound, now called benzoic acid. Surprisingly, products that appeared to retain many of the double bonds were obtained, and these compounds exhibited a high degree of chemical stability compared with known alkenes and cycloalkenes (aliphatic compounds). Since double bonds are easily cleaved by oxidative reagents such as potassium permanganate or ozone, and rapidly add bromine and chlorine, these reactions were applied to these aromatic compounds. \)īecause of the low hydrogen to carbon ratio in aromatic compounds (note that the H:C ratio in an alkane is >2), chemists expected their structural formulas would contain a large number of double or triple bonds. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
